JCRB1063 KYSE140
Cell information
Cell type:general cells (View Pricing Information)
| JCRB No. | JCRB1063 | Cell Name | KYSE140 |
|---|---|---|---|
| Profile | Human esophageal cancer cell line. | Other Name | |
| Animal | human | Strain | |
| Genus | Homo | Species | sapiens |
| Sex | M | Age | 54 |
| Identity | available | Tissue for Primary Cancer | esophagus |
| Case history | esophageal cancer | Metastasis | |
| Tissue Metastasized | Genetics | modal chromosome no.=118XX, p53 mutation in exon 6, codon 193 (CAT->CGT). | |
| Life Span | infinite | Crisis PDL | |
| Morphology | fibroblast-like | Character | |
| Classify | tumor | Established by | Shimada,Y. et.al. |
| Registered by | Shimada,Y. | Regulation for Distribution | |
| Comment | Year | 2003 | |
| Medium | F12 medium with 2% fetal calf serum. | Methods for Passages | trypsin-EDTA |
| Cell Number on Passage | Race | Japanese | |
| CO2 Conc. | 5 % | Tissue Sampling | esophagus |
| Tissue Type | moderately differentiated squamous cell carci |
| Detection of virus genome fragment by Real-time PCR | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Detected DNA Virus | tested | Detected RNA Virus | tested | ||||||
| CMV | - | parvoB19 | - | HCV | - | HTLV-1 | - | ||
| EBV | - | HBV | - | HIV-1 | - | HTLV-2 | - | ||
| HHV6 | - | HTLV-1 | - | HIV-2 | - | HAV | - | ||
| HHV7 | - | HTLV-2 | - |
-/negative. +/positive. nt/not tested. (positive (+) does not immediately mean the production of infectious viral particles.) |
|||||
| BKV | - | HIV-1 | - | ||||||
| JCV | - | HIV-2 | - | ||||||
| ADV | - | HPV18 | - | ||||||
| Notes | |||||||||
| Images |
|---|
       |
| Movies |
|---|

|
LOT Information
Viability/Growth rate/Cell number are represented as actual values measured at lot presentation in JCRB, but are not guaranteed values. Additionally, the doubling time is a rough value measured during passages.| Cell No. | JCRB1063 | Cell Name | |
|---|---|---|---|
| LOT No. | 09042006 | Lot Specification | distribution |
| Medium | Ham's F12 medium with 2% fetal bovine serum (FBS lot; GIBCO 4218333S) | Temperature | 37 C |
| Cell Density at Seeding | 1.2 - 1.8 x 10^5 cells/ml | Methods for Passages | 0.25% trypsin and 0.02% EDTA |
| Doubling Time | approx. 2 days | Cell Number in Vial (cells/1ml) | 2 x 10^6 |
| Viability at cell freezing (%) | 92.5 | Antibiotics Used | free |
| Passage Number | Unknown (10 at bank) | PDL | |
| Sterility: MYCOPLASMA | - | Sterility: BACTERIA | - |
| Sterility: FUNGI | - | Isozyme Analysis | Confirmed as human by NP, G6PD, AST. |
| Chromosome Mode | Chromosome Information | ||
| Surface Antigen | DNA Profile (STR) | ||
| Adhesion | Yes | Exoteric Gene | |
| Medium for Freezing | 10% DMSO, 20% FBS - Ham's F12 | CO2 Conc. | 5% |
| Viability immediately after thawing (%) | Additional information |
| Cell No. | JCRB1063 | Cell Name | KYSE140 |
|---|---|---|---|
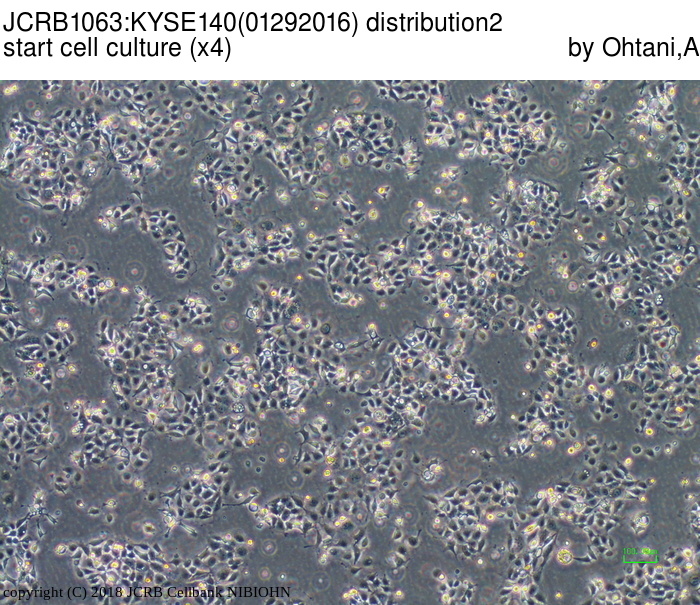
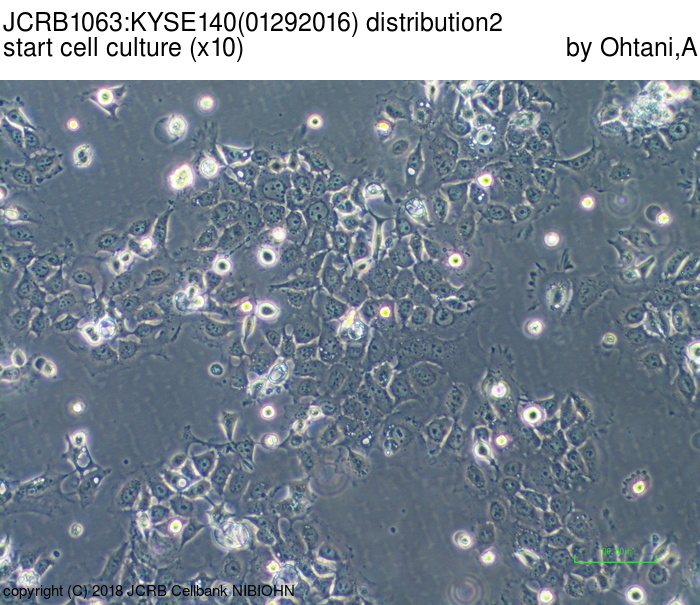
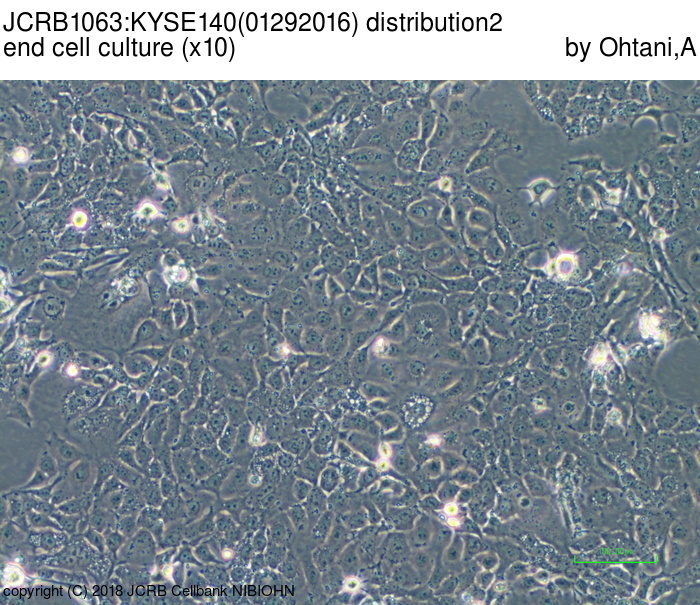
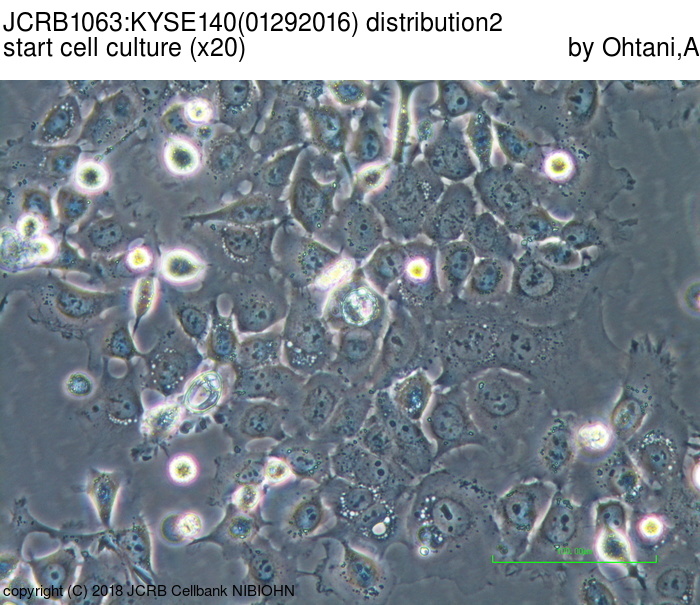
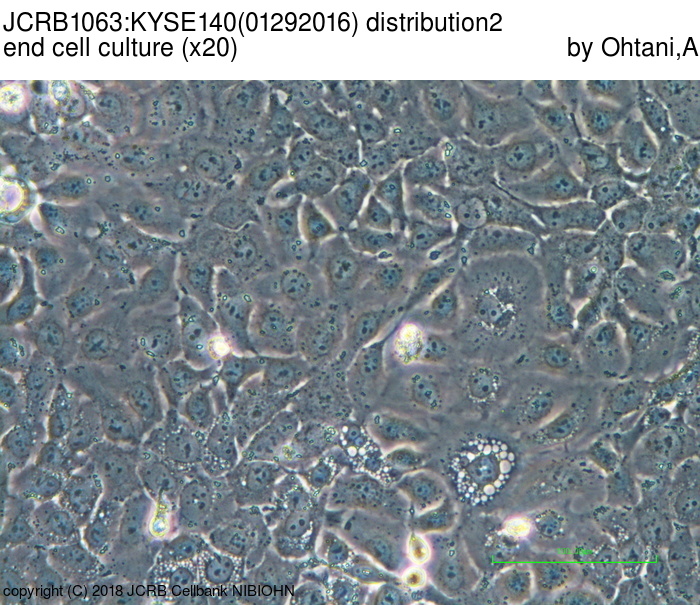
| LOT No. | 01292016 | Lot Specification | distribution2 |
| Medium | Ham's F12 medium(SIGMA) with 5% heat inactivated fetal bovine serum(SIGMA12J396) | Temperature | 37 C |
| Cell Density at Seeding | 1.39-3.13x10^4 cells/sq.cm | Methods for Passages | Cells were harvested after treatment with 0.25%trypsin(GIBCO) and 0.02% EDTA. |
| Doubling Time | NT | Cell Number in Vial (cells/1ml) | 1.72x10^6 |
| Viability at cell freezing (%) | 98.0 | Antibiotics Used | free |
| Passage Number | p14* | PDL | |
| Sterility: MYCOPLASMA | - | Sterility: BACTERIA | - |
| Sterility: FUNGI | - | Isozyme Analysis | NT |
| Chromosome Mode | NT | Chromosome Information | NT |
| Surface Antigen | NT | DNA Profile (STR) | D5S818:10 D13S317:12 D7S820:10,11 D16S539:10,12 VWA:14 TH01:7,9 AM:X TPOX:8 CSF1PO:13 |
| Adhesion | Yes | Exoteric Gene | NT |
| Medium for Freezing | BAMBANKER(LYMPHOTEC Inc.,CS-02-001,NIPPON Genetics Co., LTD) | CO2 Conc. | 5 % |
| Viability immediately after thawing (%) | 98 | Additional information | Mycoplasma and Acholeplasma has eliminated with Plasmocin(Final concentration 25 ug/ml). |
| Images |
|---|
      |
| Cell No. | JCRB1063 | Cell Name | KYSE140 |
|---|---|---|---|
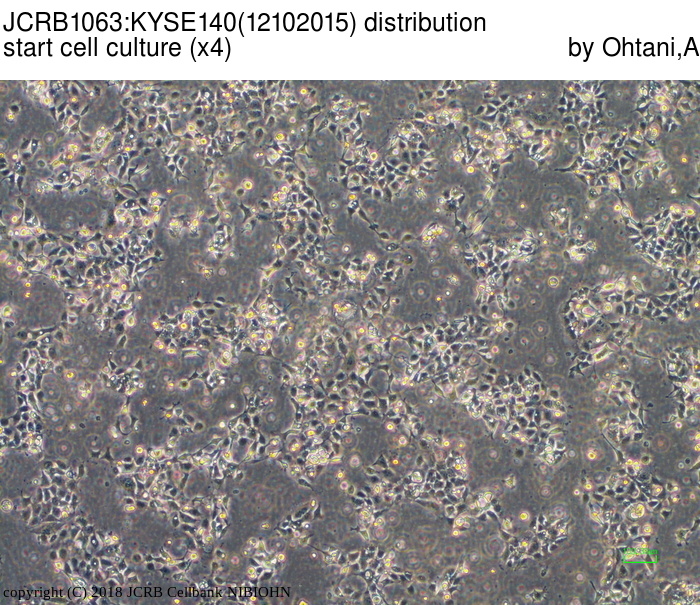
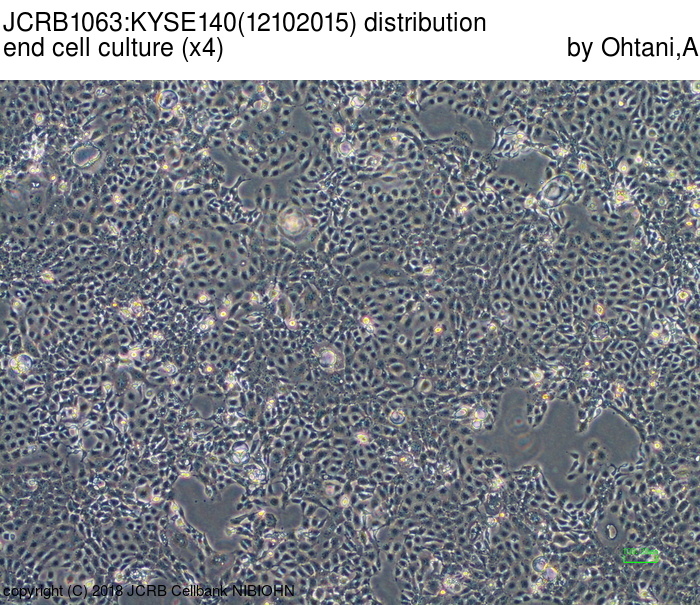
| LOT No. | 12102015 | Lot Specification | distribution |
| Medium | Ham's F12 medium(SIGMA) with 5% heat inactivated fetal bovine serum(SIGMA12J396) | Temperature | 37 C |
| Cell Density at Seeding | 1.0-1.87x10^4 cells/sq.cm | Methods for Passages | Cells were harvested after treatment with 0.25%trypsin(GIBCO) and 0.02% EDTA. |
| Doubling Time | NT | Cell Number in Vial (cells/1ml) | 1.51x10^6 |
| Viability at cell freezing (%) | 92.00 | Antibiotics Used | free |
| Passage Number | p12* | PDL | |
| Sterility: MYCOPLASMA | - | Sterility: BACTERIA | - |
| Sterility: FUNGI | - | Isozyme Analysis | NT |
| Chromosome Mode | NT | Chromosome Information | NT |
| Surface Antigen | NT | DNA Profile (STR) | D5S818:10 D13S317:12 D7S820:10,11 D16S539:10,12 VWA:14 TH01:7,9 AM:X TPOX:8 CSF1PO:13 |
| Adhesion | Yes | Exoteric Gene | NT |
| Medium for Freezing | CO2 Conc. | 5 % | |
| Viability immediately after thawing (%) | 79 | Additional information | Mycoplasma and Acholeplasma has eliminated with Plasmocin(Final concentration 25 ug/ml). |
| Images |
|---|
  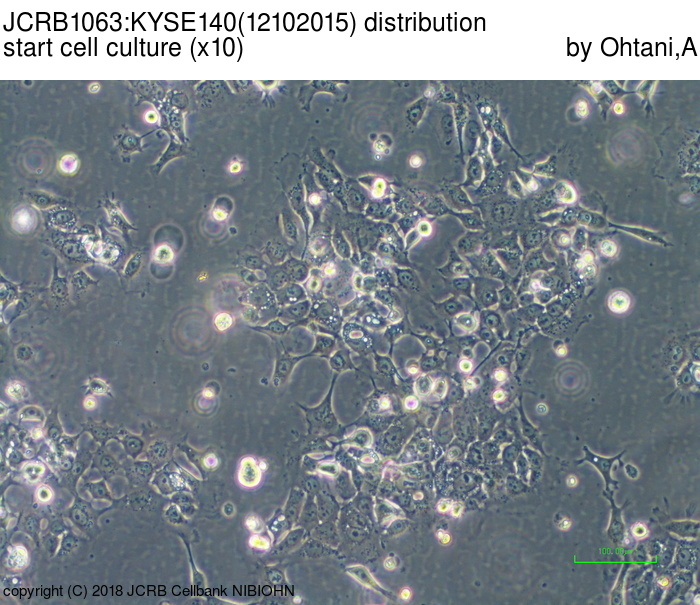 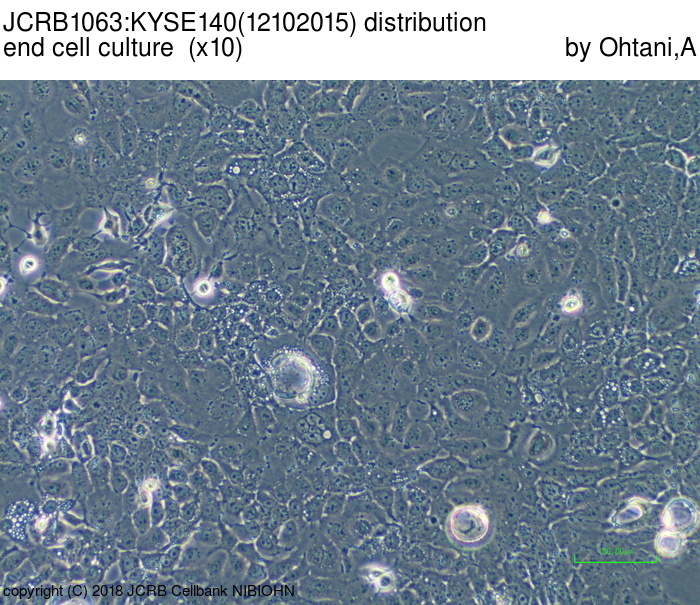 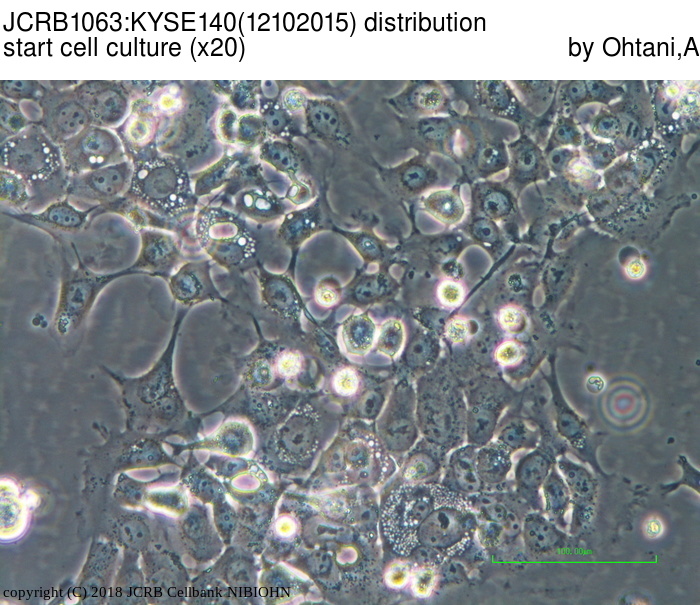 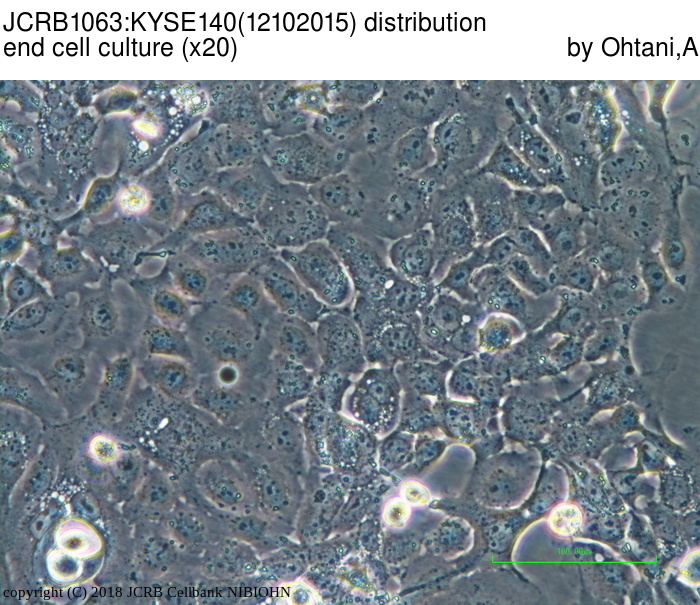 |